1-4: Atmospheric composition, carbon dioxide
Eugene S. Takle
© 1996
 RealAudio version of the learning unit
RealAudio version of the learning unit
|
One of the first indications that humans have altered the composition of
the global atmosphere comes from the measurements of atmospheric carbon
dioxide. The accompanying plot shows that from 1973 to 1985, carbon dioxide
concentrations, expressed in parts per million by volume, in the earth's
atmosphere have increased from 320 to 350. More recent measurements now
show values in excess of 350 parts per million. The plot shows that the upward
trend is independent of location and occurs in both the Northern and Southern
Hemispheres. The main differences in the curves are the different amplitudes
of the annual cycle. Careful examination shows that the concentration reaches
a peak in April or May in the Northern Hemisphere and a minimum in July,
corresponding to the absorption by plants during the growing season and the net
release back to the atmosphere due to decaying vegetation outside the growing
season. A look at the global vegetation maps for January and July will allow
you to explain the different amplitudes at different locations on the earth.
However, regardless of location, there is compelling evidence that the trend in
concentrations is upward.
|
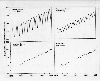 Atmospheric CO2 concentrations at Barrow, Ak, Mauna Loa, Hi,
American Samoa, and South
Pole.
Atmospheric CO2 concentrations at Barrow, Ak, Mauna Loa, Hi,
American Samoa, and South
Pole.
(1990: American Scientist, 78, 325. Permission granted
by Sigma Xi, The
Scientific Research Society.)
|
|
If we examine proxy data, we can get very good estimates of the carbon
dioxide levels over longer periods of time. At the time of the industrial
revolution in the late 1700s, the amount of carbon dioxide in the earth's
atmosphere was about 270 parts per million. The record showed that
concentrations grew slowly until the 20th century but have grown very rapidly
since then, particularly in the last 50 years.
|
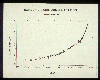 Atmospheric CO2 since
the beginning of the Industrial Revolution.
Atmospheric CO2 since
the beginning of the Industrial Revolution.
(EPA.)
|
|
We now have the capability of looking at an even longer record by
examining ice cores taken from a 2-km
deep hole drilled in the Antarctic ice sheet. A team of scientists from France
and the former Soviet Union analyzed tiny bubbles of air trapped in this core at
various levels below the present ice surface. Deeper layers correspond to times
in the more distant past. These bubbles can be analyzed for the relative
abundance of carbon dioxide to estimate atmospheric carbon dioxide levels at
times extending back 160,000 years. Concentrations of isotopes of oxygen
trapped in these bubbles are also measured, and their ratio, being temperature
dependent, gives an estimate of the surface temperature on the Antarctic
continent at the time the ice was formed. The accompanying graph shows that
carbon dioxide concentrations have gone from about 180 parts per million up to
about 300 over the last 160,000 years. And you can see from our previous
diagram also plotted here, that present concentrations exceed 350 parts per
million, higher than it's been in the last 160,000 years.
|
 Changes in global atmospheric CO2 and global surface temperature over the
last 160,000 years.
Changes in global atmospheric CO2 and global surface temperature over the
last 160,000 years.
(U.S. Global Change Research Program.)
|
|
Our discussion of magnitudes of carbon dioxide emissions on a global
scale requires a system of units that we're not accustomed to using for everyday
purposes. The accompanying table defines the prefixes, such as kilo- and
mega- that we normally use, and other to terms that may be unfamiliar. You
may find it useful to return to this table to refresh your memory on the meaning
of tera-, peta-, or exa-. I have given a couple of conversions to help
conceptualize the magnitudes of these numbers. One gigaton is the equivalent
of one petagram, and one gigaton of water is the equivalent of about one cubic
kilometer.
|
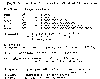 Definitions for Discussing Global Quantities.
Definitions for Discussing Global Quantities.
|
Trace gas concentrations will be given in parts per million by volume
(ppmv), parts per billion by volume (ppbv), or parts per trillion by volume
(pptv). One ppmv means one molecule out of a million. Occasionally,
concentrations are reported in parts per million by mass, but such usage will be
avoided in this course to retain uniformity.
|
We know the reason carbon dioxide is increasing in the earth's
atmosphere. Deforestation is one of these causes, although not the main cause.
The accompanying pie chart shows the current annual release to the atmosphere
of teragrams of carbon by several different countries due to deforestation. Brazil contributes the
most, followed by Indonesia, Columbia, and other mostly tropical countries. It
is important to recognize that developed countries, like the US and European
nations do not appear on the chart of current deforestation. These countries
have, in fact, reduced their forest area, but did so over the last few hundred
years |
 Net release of carbon from tropical deforestation.
Net release of carbon from tropical deforestation.
(1989: EPA, Policy Options for Stabilizing
Global Climate.)
|
|
Another source of carbon dioxide in the earth's atmosphere is the
emission from cement plants. Carbonaceous material used for making cement
releases significant amounts of carbon dioxide in creating the final product.
This source of carbon dioxide will come up again in our discussion of
sustainable development, since cement for roads, bridges, buildings, power and
manufacturing plants is a key ingredient for economic development. Of
particular interest are countries such as China and India that have such
enormous potential for development.
|
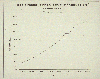 Carbon dioxide emissions
from cement production.
Carbon dioxide emissions
from cement production.
(1989: EPA, Policy Options for Stabilizing
Global Climate.)
|
|
The major cause, by far, for the rise in concentrations of carbon dioxide in the earth's atmosphere is
the burning of fossil fuels. The accompanying graph shows the fossil fuel
emissions in gigatons per year as a function of time. Emissions before the
1860s were well below 1 gigaton per year and rose quite slowly until the
middle of the present century. Since the late 1940's and early 1950's, this curve
has risen dramatically to present emission levels of about 6 gigatons per
year.
|
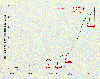 Fossil fuel emissions.
Fossil fuel emissions.
(1990: American Scientist, 78, 310. Permission granted by
Sigma, Xi, The Scientific Research Society.)
|
|
A break-down of the kinds of fossil fuels contributing to this rise shows that
coal use has increased steadily over the last 100 years and is increasing quite
significantly at present. Use of oil for heating, manufacturing and automobiles
started later but also is increasing at a rapid rate. Use of all types of
fossil
fuels is increasing, and this trend will likely continue well into the future.
|
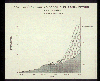 CO2 emissions due to fossil fuel consumption.
CO2 emissions due to fossil fuel consumption.
(EPA.)
|
To understand the human impact of increasing atmospheric carbon
dioxide, we first must consider the natural reservoirs and fluxes of carbon in
the earth/atmosphere/ocean system. We must include inventories of the carbon
stored in terrestrial plants and animals, carbon in the ocean, carbon in the
atmosphere, and reserves of fossil carbon that represent terrestrial carbon
taken
out of the biosphere at times in the distant past. Each of these reservoirs
takes
up and releases carbon at different rates that must be estimated to put
anthropogenic emissions into perspective.
|
The accompanying sketch shows these reservoirs, with amounts given in
petagrams of carbon and fluxes in units of petagrams of carbon per year. So,
for instance, the atmosphere contains about 740 units of carbon. Terrestrial
biology, including all of the plants from phytoplankton to giant sequoia trees
and animals from mice to elephants, accounts for about 550 units. Even these
crude numbers allow us to make simple evaluations. For instance, if humans
carried deforestation to the ultimate, in other words, if we incinerated the
whole
terrestrial biosphere and put this carbon in the atmosphere, we would
approximately double the amount of carbon dioxide in the earth's
atmosphere.
|
 Seven box schematic of the
carbon cycle.
Seven box schematic of the
carbon cycle.
(NASA.)
|
Soil contains molecular carbon, carbon of organisms living in the soil,
and detritus (broken parts of dead plants, corn stalks, and tree leaves, etc.)
in
total amount of about 1200 units. This suggests that the soil contains about
twice as much carbon as the terrestrial biology. The ocean presents a more
complicated problem for evaluating carbon stores and fluxes. The deep ocean
has about 34,000 units, and the surface ocean has about 600 units in what is
considered the warm ocean and 300 units in the cold ocean. It should be noted
that the deep ocean has a very large reservoir of carbon, but it's essentially
stored there permanently, since the cycling time from the deep ocean is on the
order of hundreds to thousands of years.
Near the ocean surface, turbulent motions promote the uptake of
atmospheric carbon dioxide by the ocean through the formation of weak
carbonic acid. Approximately 22 units per year go from the cold ocean into the
atmosphere, and about 35 units come back, making the cold ocean a net sink
for carbon dioxide from the earth's atmosphere. The warm ocean, by contrast,
is a net source because it's emitting 80 units and taking in only 70, giving a
net
flux outward of about 10 units.
Terrestrial (land) biology consumes about 110 units per year, mostly in
support of terrestrial plant growth. About 50 units of that goes back into the
atmosphere in the decay process, and about 60 units goes into the soil. The
soil, it turn, releases about 60 units back to the atmosphere, resulting in a
balance of fluxes in and out of the natural terrestrial biological system.
If we now consider the effects of anthropogenic changes to this natural
cycle, we must focus on the two largest contributions, namely burning of fossil
fuels and deforestation. Fossil fuels represent terrestrial carbon that has
been
taken out of the rapidly changing part of the carbon cycle and stored more or
less permanently below the earth's surface where natural processes cannot
release it back to the atmosphere. Fossil fuels account for about 5 to 6 units
of emission into the atmosphere, and the burning of standing carbon in the form
of old growth tropical forests contributes about 1 to 2 units.
The carbon cycle includes other carbon-containing compounds in addition
to carbon dioxide. For instance, methane molecules, which have one carbon
atom and 4 hydrogen atoms, may be produced rather than carbon dioxide in the
decay process, particularly in moist soils, marshes, and boreal tundra. Methane
also is produced by ruminant animals, such as cattle and sheep. These animals
eat large amount of grain or forage, which goes into the first of 4 stomachs
known as the rumen. The digestion process produces significant amounts of
methane that are released to the atmosphere. Carbon monoxide also is released
to the atmosphere from the burning of fossil fuels and from decay of plant
material. Volcanoes are natural sources of both carbon dioxide and carbon
monoxide.
As was previously pointed out, the ocean is both a major source and sink
of carbon for the atmosphere. Inorganic carbon in the form of dissolved carbon
dioxide and carbonates accounts for about 37,000 gigatons. Dissolved organic
carbon contributes an additional 1,000 gigatons. Particulate carbon such as
from live organisms or dead plants and animals or fragments add about 30
units. We should remember that there are large uncertainties in these estimates
because of the wide variations over the planetary oceans.
Oceans regulate carbon in 3 different ways: by physical processes,
chemical processes, and biological processes. Physical processes include the
movement of carbon by ocean circulation from one location to another.
Meteorologists and oceanographers call this process advection. Differences in
temperature and salt content (salinity), in addition to the driving force of the
wind and rotation of the earth, lead to bulk transport of carbon within and
between major ocean basins. Another physical process is the diffusive mixing
of water from one vertical level to another. Carbon dioxide dissolved in
surface water is in equilibrium with CO2 in the atmosphere because of efficient
mixing in the ocean surface water.
Chemical processes transform carbon among different molecular forms.
Biological processes include the production and decomposition of organic
matter, which are confined to the upper layer of the ocean where photosynthesis
can operate. If this biological material remains near the surface, it will
continue to cycle with the atmosphere. Some carbon, such as in the form of
phytoplankton that thrive in the surface water of the ocean, are eaten by small
fish and eventually larger fish or animals that ultimately die, leaving
skeletons
or carbonate shells that sink to the ocean floor. This process, sometimes
referred to as "biological pumping", takes carbon from the rapidly changing
part of the cycle near the ocean's surface to the deep ocean where it may be
stored for thousands of years. The deep ocean is richer in dissolved inorganic
carbon, and the surface water has a predominance of organic carbon.
The increase of ultraviolet radiation, which we will discuss in connection
with ozone depletion, also has implications for ocean biology. Many of the
simple organisms in the ocean surface are very vulnerable to ultraviolet
radiation, and increases in ultraviolet radiation due to ozone depletion could
significantly affect simple organisms at the bottom of the food chain that
thrive in the ocean surface water. This presents a linkage between the ozone
depletion problem and the build-up of atmospheric CO2: decreased ozone could
contribute to increased ultraviolet light, which decreases ocean plant life,
which
decreases CO2 consumption by the ocean, which allows for increased rates of
CO2 build-up in the atmosphere.
|
Sunlight penetrating the ocean surface is depleted as it passes downward,
creating what is called the euphotic zone where sunlight is sufficiently intense
to promote photosynthesis. In the region below the euphotic zone, the net
photosynthetic rate is negative due to lack of solar energy, resulting in very
little biological activity below a certain level. The ingredients in addition to
sunlight that are needed for biological production are nutrients, particularly
nitrogen and phosphates. The accompanying plot shows a typical nutrient
deficiency in the surface layer due to consumption by micro-organisms. Deeper
layers, where photosynthesis is suppressed due to lack of light, tend to have
elevated levels of nutrients. If a mechanism were available to bring deep,
nutrient-rich water in the euphotic zone, phytoplankton and algae would
flourish, as would the marine life that live on these tiny organisms.
Large-scale
ocean circulation patterns in certain geographical regions and vertical motions
in the ocean near continents circulate nutrient-rich water to provide the
necessary nourishment for the euphotic zone.
|
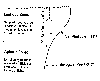 Euphotic and Aphotic Zones.
Euphotic and Aphotic Zones.
|
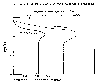 Essential nutrients for ocean
plants.
Essential nutrients for ocean
plants.
|
|
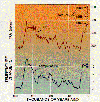
The accompanying satellite picture shows ocean biological activity in the
vicinity of Antarctica during the Southern Hemisphere spring. The color
coding indicates the level of biological activity, going from a magenta, which
represents essentially no biological activity, to blue, yellow, green, and
finally
red, which represents the highest observed concentration of phytoplankton. The
arrival of sunshine to the arctic region in the spring and early summer leads to
a rapid "bloom" of phytoplankton in this region. Notice that the regions of
highest biological activity are along continental coastlines and around the
Antarctic Continent. Equatorial ocean areas far from continents, on the other
hand, are virtual biological deserts by comparison. Certainly sunlight is not
lacking in these areas, so we must conclude that lack of nutrients prevents
these
regions from becoming biologically productive. Upwelling near continents
creates rich biological regions, but closer examination reveals that not all
coastal
areas are equally productive. Again, we can conclude that differences in
upwelling of nutrient-rich water must be the cause of these differences in ocean
biology.
|
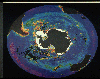 Biological activity in the
oceans of the southern hemisphere.
Biological activity in the
oceans of the southern hemisphere.
(July/Aug 1990: American
Scientist.
Permission granted by Sigma Xi, The Scientific
Research Society.)
|
|
The next photograph of the Northern Hemisphere, centered on the North
Pole shows that the whole North Atlantic Ocean is a very biologically productive
region. In the North Pacific Ocean, coastal regions between Alaska and Russia
also show high levels of phytoplankton. Considering that these small organisms
cover such a large area and consume CO2 , we must conclude that the polar
oceans are tremendous sinks (removal mechanisms) for atmospheric carbon
dioxide. In contrast to lower latitudes where lack of nutrients limits
biological
activity except near coasts, in the polar regions the supply of nutrients is
persistent, but the lack of sunlight during winter periods shuts down the
phytoplankton production beginning in autumn. However the large extent of
the summer blooms means that a tremendous amount of carbon dioxide is used
seasonally by these organisms. A closer look at coastal California in the next
figure shows that northern California experiences a rich phytoplankton bloom,
which is less pronounced in the ocean off southern California. Ocean currents
(to be discussed later in the course) produce upwelling preferentially in the
northern part of the state, a situation that also is responsible for ocean
temperatures off San Francisco being much colder than those off Los Angeles.
|
 Biological activity in the
oceans of the northern hemisphere.
Biological activity in the
oceans of the northern hemisphere.
(July/Aug 1990: American Scientist.
Permission granted by Sigma Xi, The Scientific
Research Society.)
|
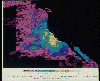 Mean near-surface
phytoplankton pigment concentrations off the California coast. (NASA.)
Mean near-surface
phytoplankton pigment concentrations off the California coast. (NASA.)
|
A limiting factor for the growth of phytoplankton and algae around
Antarctica, even during the spring bloom, is insufficient amounts of iron. A
proposal has been made that by fertilizing the Antarctic ocean with iron, the
growth in ocean marine plants would sequester large additional amounts of
carbon dioxide from the atmosphere and counteract the anthropogenic increases.
More information on this "iron solution" is given on the
electronic dialog,
where you are encouraged to voice your opinion on the wisdom of such
"engineering approaches" to solving environmental problems.
|
Terrestrial plants consume carbon dioxide during the daytime period and
respire some of this CO2 back to the atmosphere at night. The
accompanying
sketch shows typical levels of CO2 within a vigorously growing plant canopy at
different times of day and night. At night, plants tend to respire or give up
carbon dioxide, raising the ambient level well above the global mean (which was about 330 ppm when this sketch was printed).
During the day however, if the plants are not limited by lack of water or
nutrients, photosynthesis may draw down the amount of CO2 to approximately
300 parts per million. The carbon taken up by the plants is converted to plant
carbon and remains in this form until the decay process begins. For typical
agricultural plants, the biomass is broken down during the dormant period and
is converted to soil carbon and atmospheric carbon dioxide at rates that depend
on temperature and moisture conditions.
|
 Vertical distribution of
carbon dioxide in the air around a forest varies with time of day.
Vertical distribution of
carbon dioxide in the air around a forest varies with time of day.
J. D. Butler, Air Pollution Chemistry, 1979.
|
Woody plants play a special role in the capture (sequestration) of carbon
from the atmosphere. Trees lose their leaves (deciduous) and some or all
needles (conifers) every year and return a portion of the season's carbon
capture back to the atmosphere. The woody parts of the tree, however, may
persist as plant carbon for several decades until the tree dies and decay
begins.
Trees, therefore, like annual plants, participate in the rapid part of the
carbon
cycle by cycling CO2 back to the atmosphere in 1-3 years, but also store some
carbon that is not recycled to the atmosphere for 50 to 100 years. Tropical
rain forests have very large amounts of carbon sequestered in the trunks of trees
as a part of this longer term cycle. Deforestation interrupts this natural long
term cycle and puts woody material back into the atmosphere before the natural
decay process would recycle it into CO2 .
|
The "greenness index" of the planet, shown in the accompanying figure
for July and August shows very rich terrestrial biological activity in the
Northern Hemisphere continental areas, particularly in the boreal forests of
northern Russia and Canada. At this time of year, the Southern Hemisphere, of
course, is experiencing the biologically dormant winter period. Major desert
regions, located approximately 30o north and south of the equator, show lack of
vegetation in both hemisphere (both summer and winter). During the Northern
Hemisphere winter, shown in the next figure, the boreal forest regions are seen
to have shut down their biological production. In the Southern Hemisphere the
smaller amount of land mass does not reveal the dramatic seasonal variation of
the Northern Hemisphere. This suggests that global seasonal cycles in
atmospheric carbon dioxide will be dominated by the seasonal cycle in the
Northern Hemisphere.
|
 July-August global plant biological activity as determined by the Global Vegetative Index.
July-August global plant biological activity as determined by the Global Vegetative Index.
American Scientist. Permission granted by Sigma, Xi, The Scientific Research
Society.
|
 January-February global plant biological activity as determined by the Global Vegetative Index.
January-February global plant biological activity as determined by the Global Vegetative Index.
American Scientist, 78, 322 (1990). Permission granted by Sigma, Xi, The Scientific
Research Society.
|
|
The accompanying figure shows biological production potential for the entire
planet, including both ocean and land areas. An important conclusion to be
drawn from this figure is that relatively large regions of the planet, including
tropical oceans, great deserts on land, and Antarctica, have very low potential
for biological production. Essentially all the biologically productive land
that is
not in forests or urban use is under human agricultural management, and most
of the productive ocean areas are heavily fished by global fishing fleets from
many countries. Increased food production to feed a growing population on
the planet will require higher production on present agricultural land or more
conversion of forest land to agricultural land, both of which present problems
and challenges. This figure reminds us of the finiteness of our planet and the
need for preservation and stewardship of its food-producing resources.
|
 Productive potential of the
Earth's vegetative biomass.
Productive potential of the
Earth's vegetative biomass.
(NASA)
|
Transcription by Theresa M. Nichols

![]() RealAudio version of the learning unit
RealAudio version of the learning unit July-August global plant biological activity as determined by the Global Vegetative Index.
July-August global plant biological activity as determined by the Global Vegetative Index.
 January-February global plant biological activity as determined by the Global Vegetative Index.
January-February global plant biological activity as determined by the Global Vegetative Index.